The fascinating universe of fishkeeping can pose a significant challenge for beginners, who frequently encounter a troubling proliferation of misinformation. One of the most critical—and often misunderstood—biological pillars is aquarium cycling. The recurring narrative of introducing fish on the very day of setup, only to find them lifeless by the next morning, serves as empirical evidence of what is known as “New Tank Syndrome.”
In this comprehensive technical guide, we will rigorously analyze the biochemical processes involved. We will explore everything from methodologies for optimizing maturation times to the critical distinctions between fishless cycling, fish-in cycling, and planted cycling. Prepare to understand and master water chemistry with precision to ensure a healthy ecosystem for your fish.
- 1 Key Takeaways: Essential Facts of Aquarium Cycling
- 2 What is Aquarium Cycling and Why is it Vital?
- 3 Advanced Microbiology: The Invisible Protagonists
- 4 The Science of Surface Area: Biofilm and Bacterial Infrastructure
- 5 How to Cycle a New Aquarium: Methods and Strategies
- 6 Strategies for Accelerating the Cycle: Proven Biological “Shortcuts”
- 7 The Great Divide: Commercial Products vs. Scientific Reality
- 8 The Final Verification Protocol
- 9 The Concept of a “Mature Aquarium” vs. “Basic Cycling”
- 10 Technological Trends in Biofiltration: The Future of Aquaria
- 11 Conclusion: Cycling as Life Insurance
-
12
Aquarium Cycling Frequently Asked Questions (FAQ)
- 12.1 How long does it actually take to cycle an aquarium?
- 12.2 Can I cycle my aquarium in just 24 hours with commercial products?
- 12.3 Why did my aquarium water turn white or cloudy during the second week?
- 12.4 Is a water change necessary during cycling?
- 12.5 What is the difference between a “cycled” and a “mature” aquarium?
- 12.6 Why do my plants stall the cycling process in the filter?
- 12.7 What is the best filter media for bacteria?
- 12.8 Why does the Walstad method discourage heavy aeration?
- 13 References
- 14 Entradas relacionadas:
Key Takeaways: Essential Facts of Aquarium Cycling
- Cycling is Mandatory: It is the biological process of establishing nitrifying bacteria (Nitrosomonas and Nitrospira) that convert toxic ammonia into harmless nitrates. Without this, “New Tank Syndrome” occurs—the leading cause of fish mortality.
- Evolving Science: Recent studies demonstrate that comammox Nitrospira and ammonia-oxidizing archaea (AOA) are the true drivers of a mature biofilter, superseding the traditional bacteria cited in older literature.
- The “Gold Standard” is Fishless Cycling: This is the most ethical and secure method. It allows for controlled ammonia levels of 2 to 4 ppm, fostering a robust bacterial colony without risking animal life.
- Cycling vs. Maturation: An aquarium is “cycled” within weeks (chemically stable), but it is only “mature” after 3 to 6 months of operation, once it develops a complex biofilm and resilient microfauna.
- The Importance of Specific Surface Area (SSA): Not all filter media are created equal. Sintered glass and porous ceramics offer up to 10 times more colonization area than sponges, enabling a more stable ecosystem in less space.
- The Role of Plants: In the Walstad Method, plants (especially floating species) act as an instantaneous biological filter by preferring ammonia as a nitrogen source, “silently” reducing toxic spikes.
- Beware of Commercial “Shortcuts”: Many bacterial supplements fail to reduce ammonia within the first 14 days. Use them as support, but never rely on them blindly without performing liquid drop tests.
- The Aquarist’s Golden Rule: In fishkeeping, only negative things happen quickly. Patience and constant monitoring with precision kits are your best tools for long-term success.
What is Aquarium Cycling and Why is it Vital?
Cycling is the biological process of establishing a colony of nitrifying bacteria (primarily of the genera Nitrosomonas and Nitrospira) within the filtration system. Technically, it consists of instating the nitrogen cycle via biological filtration in a closed ecosystem: we cultivate beneficial microorganisms that metabolize toxic fish waste, transforming it into assimilable or less harmful compounds.
The so-called “New Tank Syndrome” represents the primary cause of mortality in fishkeeping. This phenomenon occurs when introducing fauna (fish, shrimp, or other aquatic organisms) into an environment lacking an active microbiota. Without these bacteria, waste accumulates as pure ammonia—a caustic compound that erodes gills and collapses the nervous system of specimens within hours.
Editorial Note: Cycling an aquarium is not about “cleaning the water,” but rather about fostering a microscopic life infrastructure that functions as an invisible waste treatment plant.
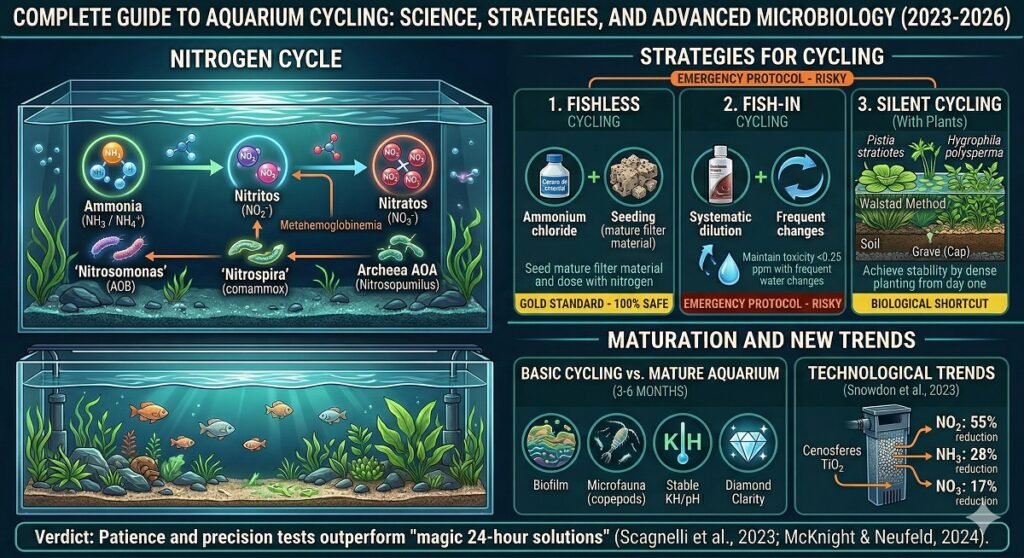
The Nitrogen Cycle: The Chemistry Behind the Glass
To successfully manage an aquarium, it is imperative to understand nitrogen biochemistry. In nature, this cycle occurs organically on a large scale; in captivity, we must artificially recreate it through three critical phases:
- Ammonia ( / ): Generated through branchial excretion, organic debris, and decomposing plant biomass. It is highly cytotoxic; even in minute concentrations, it causes chemical burns to fish tissues.
- Nitrites (): Nitrosomonas bacteria process ammonia and excrete it as nitrite. Although an intermediate metabolite, its toxicity is extreme, as it induces methemoglobinemia, preventing the fish’s blood from effectively transporting oxygen.
- Nitrates (): Finally, bacteria such as those of the genus Nitrospira convert nitrite into nitrate. This compound is significantly less toxic, and its accumulation is managed through partial water changes or absorption by live plants.
Advanced Microbiology: The Invisible Protagonists
To achieve the technical depth required by modern fishkeeping, we must transcend generic labels. The nitrification process is primarily executed by obligate chemolithoautotrophs—microorganisms specialized in deriving energy from inorganic compounds.
Nitrosomonas: Specialists in Ammonia Oxidation
These bacteria oxidize ammonia () into nitrite () in an exergonic process used for carbon fixation. A critical factor is their dependence on inorganic carbon (carbonates). In waters with excessively low carbonate hardness (KH), cycling may stall due to the lack of this essential “building block.”
The Enigma of Nitrospira vs. Nitrobacter
Historically, the conversion of nitrite to nitrate was attributed to Nitrobacter. However, contemporary phylogenetic analyses confirm that in home aquaria, the genus Nitrospira is the true biological engine. According to Elmagzoub et al. (2026), bacteria from the phylum Nitrospirae are decisive for ecosystem stability:
- Establishment and Detoxification: They are vital components in bacterial inocula, processing ammonia derived from biological excretion.
- Dynamic Response: They possess a remarkable capacity for population expansion during sudden increases in organic load (such as the introduction of new fish or filter media changes), representing up to 40% of the bacterial community following management interventions.
The New Paradigm: Archaea and Comammox Bacteria
Vanguard research has revealed players that previously went unnoticed:
Stay Always Informed
Join our communities to instantly receive the most important news, reports, and analysis from the aquaculture industry.
- Ammonia-Oxidizing Archaea (AOA): Sauder et al. (2011) and Bagchi et al. (2014) demonstrated that AOA are numerically dominant over bacteria (AOB) in most freshwater biofilters. Surprisingly, many commercial supplements lack these archaea, despite their critical role in ammonia catalysis.
- Comammox Nitrospira Bacteria: McKnight and Neufeld (2024) highlight these organisms capable of performing complete ammonia oxidation in a single step. Toward the end of a 12-week maturation period, comammox typically establishes itself as the dominant group in freshwater systems (McKnight et al., 2025).
Critical Differences: Freshwater vs. Marine Ecosystems
Microbiological composition varies drastically based on salinity:
- Freshwater: Dominated by comammox bacteria and archaea from the families Nitrosopumilaceae and Nitrososphaeraceae (Godzieba et al., 2025).
- Saltwater: Oliaro et al. (2025) identified the genus Nitrosopumilus as the primary driver of the early cycle in marine aquaria, contradicting the traditional hypothesis that assigned this role to AOB bacteria. Interestingly, the presence of Comammox Nitrospira is not detected in marine systems (McKnight and Neufeld, 2024).
The Science of Surface Area: Biofilm and Bacterial Infrastructure
Nitrifying bacteria do not drift freely within the water column; they establish complex biological “metropolises” known as biofilms. This biofilm is a matrix of extracellular polymeric substances (a protective slime) that shields microorganisms from chemical fluctuations and predators.
Technical Warning: Cleaning filter media with chlorinated water not only eliminates bacteria but also dismantles their physical infrastructure, forcing the ecosystem to restart its development from scratch.
Filter Media Selection: Where Do Bacteria Reside?
The efficiency of biological filtration is contingent upon the material’s Specific Surface Area (SSA). Not all supports offer equivalent performance:
- Sintered Glass and High-Porosity Substrates (e.g., Seachem Matrix, Sera Siporax): These represent the technological vanguard. Their micro-tunnel structure offers up to 10 times more colonization area than conventional media and prevents clogging (sludge obstruction). According to McKnight et al. (2025), ammonia-oxidizing archaea (AOA) show an intrinsic preference for colonizing ceramic cylinders and glass beads.
- Foamex Sponges: While vital for mechanical filtration, Bagchi et al. (2014) found that AOA reach their highest abundance in fine-pored sponges, even outperforming ceramic rings in certain initial attachment contexts.
- Bio-balls: Designed for wet-dry systems (sumps). In internal or hang-on-back (HOB) filters, their efficiency is significantly lower due to their low internal porosity.
Environmental Factors: Optimizing the Biological “Laboratory”
To accelerate bacterial metabolism during cycling, it is essential to parameterize the environment:
- Temperature: Optimal growth occurs between 25°C and 30°C. In fishless cycling, increasing the heater temperature accelerates cell division.
- Oxygenation: Nitrification is a strictly aerobic process. Vigorous surface agitation is indispensable.
- Potential of Hydrogen (pH): Colonies thrive in slightly alkaline media (pH > 7.0). A drop below 6.0 can inhibit or halt nitrifying activity.
Specialized Ecology and the Future of Filtration
Research by Zhou et al. (2022) reveals that an aquarium is not a uniform mass, but a mosaic of microbiological niches:
- Plants: They host genera such as Nitrospira and Rhodobacter, being the component with the highest bacterial diversity in the tank.
- Substrate (Sand): Genera such as Pseudomonas and Rhodococcus predominate.
- Fish and Water: They present distinct microbiome profiles, dominated by Cetobacterium and Flavobacterium, respectively.
Finally, innovations like the cellulose substrate bioreactor developed by Ponpornpisit et al. (2024) suggest a paradigm shift: the capacity to perform simultaneous nitrification and denitrification in a single tank. This advancement would allow for the reduction of not only ammonia and nitrites but also nitrates, drastically simplifying long-term maintenance.
How to Cycle a New Aquarium: Methods and Strategies
To establish a robust colony of nitrifying bacteria and consolidate the nitrogen cycle, various operational protocols exist. These methodologies are primarily categorized into three fundamental approaches, each with specific technical requirements and maturation timelines:
- Fishless Cycle: The gold standard in safety, utilizing synthetic ammonia or organic matter to feed the microbiota before introducing fauna.
- Silent Cycle (Planted): An advanced ecological strategy that leverages the nitrogen-processing capacity of plant biomass.
- Fish-in Cycle: A traditional, albeit technically more demanding method, requiring exhaustive monitoring to ensure animal welfare.
Technical Comparative Table: Aquarium Cycling Methodologies
| Evaluation Criteria | Fishless Cycle | Fish-in Cycle | Silent Cycle (Planted) |
| Difficulty Level | Low: Automated process. | Critical: Requires daily rigor. | Moderate: Requires botanical knowledge. |
| Biological Safety | Maximum (100%): No animal risk. | Low: Potential stress and toxicity. | High: Natural mitigation of spikes. |
| Maturation Time | 4 to 6 weeks (Stable). | 4 to 8 weeks (Unstable). | Variable: Depends on biomass. |
| Financial Investment | Minimal: Ammonia or food. | High: Continuous tests and chemicals. | Medium: Live flora acquisition. |
| Technical Requirement | Patience and basic monitoring. | Water changes and neutralization. | Balance of light, $CO_2$, and nutrients. |
In addition to these categories, we will explore biological acceleration strategies and inoculum seeding techniques to optimize wait times and achieve ecosystem stability in record time.
Fishless Cycling: The Gold Standard
Fishless cycling is the “gold standard” in modern fishkeeping. This method involves introducing a source of synthetic ammonia or organic matter into an empty tank to foster bacterial growth without compromising animal welfare. It is the most ethical and secure strategy, as it allows for the management of toxicity levels that would be lethal to any species.
Dosing Methodologies
There are three primary techniques to supply the necessary nitrogen:
- Pure Ammonia or Ammonium Chloride: The most precise method. It allows for reaching specific concentrations (between 2 and 4 ppm) without fouling the water with organic waste.
- Ghost Feeding: Consists of adding fish food daily. As it decomposes, it releases ammonia gradually, though it is less precise and generates detritus.
- Raw Shrimp or Fish Method: A piece of shrimp or fish (protected in a mesh bag) is introduced; its decomposition provides a massive and constant source of ammonia.
Step-by-Step Protocol: From Sterile Tank to Living Ecosystem
Step 1: Setup and Conditioning
Install the aquarium and fill the tank with treated water. It is vital to remove chlorine and chloramines, as they are bactericidal. Walker and Jiang (2019) suggest using ascorbic acid to neutralize sodium hypochlorite at a 2.5:1 ratio (mg/L).
- Optimization: Raise the temperature to between 29°C and 32°C and maximize surface oxygenation to accelerate colony metabolism.
- pH Control: Ensure the pH remains above 7.0; if it drops below 6.0, nitrification may be inhibited.
Step 2: Inoculation (Optional)
To reduce the 6-week wait time to just 1 or 2 weeks, introduce live bacteria using filter media from a healthy aquarium or high-end commercial products (FritzZyme TurboStart or DrTim’s One & Only).
Step 3: Ammonia Dosing
Add the nitrogen source until reaching 2–3 ppm. According to Sauder et al. (2011), lower concentrations favor the abundance of Ammonia-Oxidizing Archaea (AOA), while higher levels promote the growth of AOB bacteria. Never exceed 5 ppm, as this becomes toxic even to beneficial bacteria.
Step 4: Analytical Monitoring
Use precision liquid drop kits (avoid test strips due to their high margin of error).
- Dynamics: You will see a nitrite () spike while ammonia levels drop.
- Microbial Succession: Oliaro et al. (2025) recommend monitoring nitrifiers as a true indicator of system readiness, as chemical parameters do not always accurately reflect microbiome stability (Bik et al., 2019).
| Phase | Ammonia (NH3) | Nitrites (NO2−) | Nitrates (NO3−) | Action Required |
| Start | 2–4 ppm | 0 ppm | 0 ppm | Dose nitrogen source |
| Mid | Decreasing | Peak | Increasing | Maintain feeding (1 ppm) |
| Final | 0 ppm | 0 ppm | > 20 ppm | Massive water change |
Step 5: Verification and Biological Load
The cycle is complete when the system can process 2–3 ppm of ammonia to 0 ppm of nitrites within 24 hours. Before introducing fish, perform a 50% to 90% water change to reduce accumulated nitrates. Fauna introduction must be gradual to avoid “mini-cycles” caused by excessive organic load.
Fish-in Cycling: The Emergency Protocol
Fish-in cycling, often referred to as an “emergency protocol,” is an exercise in extreme biochemical balancing. It involves introducing specimens (typically hardy species such as Danio rerio) so that their metabolic processes generate the initial ammonia. However, this method is discouraged for beginners, as it exposes fauna to cytotoxic conditions that can compromise their immune systems or prove lethal. If you find yourself in this situation, the primary objective shifts from accelerated bacterial growth to the systematic dilution of toxins.
Survival and Mitigation Protocol
To execute fish-in cycling technically and safely, it is imperative to follow these guidelines:
- The Dilution Rule: The key is to maintain ammonia () and nitrite () levels below 0.25 ppm. This requires daily analytical monitoring and frequent water changes (30% to 50%) whenever parameter deviations are detected.
- Organic Load Management: Extremely light feeding must be implemented. Lower organic input translates to reduced ammonia production, easing the workload for the nascent bacterial colony.
- Chemical Neutralization (Reduction Conditioners): The use of advanced conditioners, such as Seachem Prime, is indispensable. These products have the capacity to temporarily “sequester” ammonia and nitrite, converting them into non-toxic forms (stable molecular complexes) that bacteria can still metabolize without harming the fish’s gills.
Professional Advice: Fish-in cycling is not a time-saving strategy, but rather a greater technical responsibility. Without a precision testing kit and disciplined water change routine, the risk of total ecosystem loss is imminent.
Silent Cycling (Planted Cycling): The Biological Shortcut
Silent Cycling, or planted cycling, leverages the ability of higher plants to directly absorb ammoniacal nitrogen even before bacteria begin processing it. In this model, plant biomass acts as a “chemical sponge,” mitigating toxic nitrite spikes in an almost imperceptible manner. Strategies such as the Walstad Method (organic substrate under a sand cap) demonstrate that a heavily planted aquarium can achieve organic stability from day one. However, current science provides critical nuances regarding this practice:
Scientific Evidence and Limitations
- Natural Inoculation: McKnight et al. (2025) observed that aquaria with live plants achieve earlier nitrification than those with artificial decorations and commercial supplements. This suggests that plants are natural vectors for nitrifying microorganisms, potentially rendering bottled bio-supplements unnecessary.
- Microbiological Richness: According to Zhou et al. (2022), aquarium plants and their associated bacterial communities constitute the most biodiverse component of the aquarium, surpassing both water and substrate in microbial richness.
- The Performance Debate: Despite these benefits, Asadi et al. (2022) concluded that the exclusive use of plants such as Ceratophyllum demersum (Hornwort) does not necessarily outperform artificial filters in nitrogenous compound management, even noting a decrease in fish growth parameters within purely botanical systems.
Competition Dynamics and Risks
In an aquarium where 70% of the substrate is covered by flora, plants compete directly with bacteria for ammonia (), which they prefer over nitrate ().
- Advantage: Toxicity levels remain undetectable from the initial phase.
- Collapse Risk: The bacterial colony in the filter is often smaller due to this competition. If plant biomass is abruptly removed or dies due to lighting deficiencies, the system will lack a robust bacterial population to compensate for the load, triggering a lethal ammonia spike.
Recommendation: For successful cycling, use fast-growing species with high nitrogen demand, such as Pistia stratiotes (Water Lettuce) or Hygrophila polysperma. These plants act as a biological safety system while the microbiota colonizes the porous filter media.
The Walstad Method Step-by-Step: Creating a Self-Sustaining Ecosystem
The method developed by Diana Walstad seeks to recreate nature’s balance within a closed system. Unlike conventional aquaria, here, plants and organic substrate are the primary drivers of filtration.
Step 1: Organic Substrate Preparation
The core of the system is the use of organic potting soil (low in nitrogen, <0.10%, and free of chemical fertilizers).
- Process: Sift the soil to remove wood debris that could float or rot.
- Installation: Lay a 0.5 to 1-inch layer.
- Expert Tip: Adding a teaspoon of pond mud from a healthy source will inoculate invaluable microbial biodiversity from day one.
Step 2: Capping
To prevent the soil from clouding the water, seal it with a 0.5 to 1-inch layer of porous gravel or coarse sand. This “cap” allows for slight oxygen exchange, essential for cellulose decomposition to generate the $CO_2$ that will feed your plants. Lightly mist with a spray bottle to settle the layers.
Step 3: Layout and Hardscape
Add rocks or driftwood. Consider that decomposing wood consumes nitrates and phosphates; monitor if your plants require nutrient supplementation in later stages.
Step 4: Heavy Planting and the Role of Floaters
The golden rule is to plant heavily from the start. Plants are your primary filter.
- Submerged Species: Combine fast-growing plants (Ludwigia, Hornwort) with root feeders (Cryptocoryne, Echinodorus).
- The Necessity of Floaters: Species such as Salvinia, Pistia (Water Lettuce), or Limnobium laevigatum (Amazon Frogbit) are vital. Having access to atmospheric , they process ammonia at a much higher rate than submerged plants, outcompeting algae.
Step 5: Precision Filling
Pour water extremely slowly over a plate or plastic bag to avoid breaking the substrate seal and preventing soil from mixing with the water column.
Step 6: Lighting and Circulation
- The “Siesta” Cycle: To maximize natural and mitigate algae, use a schedule of 4h on / 5h off / 4h on.
- Circulation: Gentle movement (a small filter or powerhead) is recommended to distribute nutrients. Avoid heavy aeration (air stones), as it outgasses the that the soil works so hard to produce.
Step 7: Cycling and Biological Load (The Cubic Relationship)
Although the system is efficient, fish introduction must be gradual and based on oxygen availability, not just ammonia.
The Science of Consumption: Fish impact is not linear, but cubic. A 2-inch fish (like a Platy) consumes 8 times more oxygen than a 1-inch fish. In a Walstad system without aeration, oxygen is the limiting factor, especially at night when plants also consume it.
Recommended Species (Low Impact):
- Small Fish: Ember Tetras or Chili Rasboras (max 1 inch).
- Livebearers: Guppies or Endlers (preferably males only to avoid overpopulation).
- Ideal Ratio: Maintain a ratio of 10 plants for every fish. A successful 10-gallon aquarium looks like an “absolute jungle” with barely 6 small tetras.
Strategies for Accelerating the Cycle: Proven Biological “Shortcuts”
The recurring question among aquarium enthusiasts is: how can I speed up the cycling process? While the natural biochemical maturation typically ranges from 4 to 8 weeks, applied science allows us to implement acceleration strategies to drastically reduce these timelines.
Seeding with Mature Media
The most effective technique for rapid cycling is not a chemical agent, but the direct transfer of active biology. This method, known as “seeding,” involves introducing “mature” filter media (sponges, ceramic rings, or bio-balls) from a healthy, established system.
- Process Mechanics: By transferring colonized supports, billions of nitrifying bacteria ready to process nitrogen are relocated.
- Temporal Impact: This action can reduce the wait time from a month to just one week, enabling what is technically termed an “instant cycle” in optimal cases.
- Marine Equivalents: In saltwater systems, this concept is replicated through the use of high-quality live rock or live sand.
Bio-supplements and Bottled Bacteria
The use of commercial supplements containing live strains (primarily of the genera Nitrosomonas and Nitrospira) provides the system with an initial competitive advantage.
- Critical Consideration: It is essential to select professional-grade products. Some low-cost variants employ soil-based bacterial strains which, while initially processing ammonia, fail to establish themselves permanently in aquatic environments, leading to biological crashes in the medium term.
- Function: They act as a primary inoculum that accelerates the colonization of filter surfaces much sooner than spontaneous environmental propagation would allow.
Expert Pro-Tip: The most potent way to start an aquarium is to combine both methods: seed with used filter media and reinforce the population with a high-end bacterial supplement during the first week.
The Great Divide: Commercial Products vs. Scientific Reality
We now enter the most controversial territory in aquarium cycling: “bacteria in a bottle” supplements that promise a 24-hour cycle. Is this biologically feasible, or merely a marketing strategy?
The Bottled Bacteria Debate
Products across various price points coexist in the market, but microbial science establishes a fundamental distinction:
- Heterotrophic Bacteria: Found in low-cost products; they decompose organic matter (sludge) but do not fix nitrogen efficiently over the long term.
- True Autotrophic Bacteria: Only a few high-end supplements contain live strains of Nitrosomonas and Nitrospira capable of permanently colonizing filter media.
Evidence from Contemporary Research (2023–2025)
Recent scientific literature casts a skeptical light on the efficacy of these “quick-start” nitrifiers:
- Questionable Efficacy: Scagnelli et al. (2023) evaluated five quick-start nitrifying products (QSNPs). The results were revealing: 80% of the analyzed products failed to reduce Total Ammoniacal Nitrogen (TAN) concentrations over 14 days. The study recommends against relying exclusively on these products to initiate the nitrogen cycle.
- Industry Technological Lag: McKnight et al. (2025) note that since comammox bacteria and AOA archaea are relatively recent discoveries, the industry has historically based its formulas on AOB bacteria. However, research demonstrates that AOB are eventually displaced by comammox Nitrospira—the true protagonists of a mature biofilter.
- Suboptimal Formulas: Godzieba et al. (2025) suggest that traditional cultures (generally based on Nitrobacter) are not optimal for modern aquarium environments, necessitating a reevaluation of both the production and use of these commercial preparations.
Advice: These products may function as “starters” or initial catalysts, but under no circumstances do they replace the need for monitoring with liquid drop kits. The myth of “add the product and add the fish” remains the leading cause of mortality in the hobby.
The Final Verification Protocol
It is fundamental not to be misled by water clarity or the simple passage of time. The only irrefutable technical validation is obtained through precision chemical analysis. An ecosystem is officially considered “cycled” when, after introducing a nitrogen source, the following conditions are simultaneously met:
- Ammonia (): A consistent reading of 0 ppm.
- Nitrites (): A consistent reading of 0 ppm.
- Nitrates (): A detectable presence (generally between 5 and 20 ppm), confirming that the nitrification chain is complete.
The 24-Hour Stress Test: To confirm that your biological filter is functional, perform a final dose of 2 ppm of ammonia. If, within a 24-hour period, both ammonia and nitrites have returned to 0 ppm (having been fully converted into nitrate), the system is ready for the gradual introduction of fauna.
Cycling vs. Maturation: The Hobby’s Most Common Error
Most informational resources conclude their advice once nitrites reach zero. However, a newly cycled aquarium is chemically stable but biologically immature. While cycling takes weeks, comprehensive biological maturation—the development of a complex microfauna of biofilm, protozoa, and zooplankton—typically requires 3 to 6 months.
Indicators of a Mature Ecosystem:
- Active Biofilm Presence: A thin biological layer on driftwood and rocks that constitutes the nutritional base for invertebrates and microorganisms.
- KH and pH Stability: Carbonate consumption by bacteria stabilizes, preventing dangerous acidity fluctuations.
- “Diamond” Clarity: Exceptional transparency in the water column, achieved through the efficient removal of organic micro-particles by the established microbial community.
The Concept of a “Mature Aquarium” vs. “Basic Cycling”
A systemic error in fishkeeping is equating a “cycled” aquarium with a “stable” one. Cycling is merely the initial phase of bacterial colonization; true biological stability is a maturation process that requires months.
A mature aquarium has surpassed 90 days of uninterrupted operation. At this stage, a complex biofilm and auxiliary microfauna (such as copepods and protozoa) have consolidated, assisting in the processing of micro-waste. During this first trimester, the ecosystem remains extremely fragile; the mass introduction of fauna can overwhelm the young bacterial colony and trigger a biological collapse.
The Water Change Controversy: Error or Salvation?
There is an ongoing technical debate regarding the suitability of performing water changes during the maturation process. The science of nitrification establishes distinct protocols based on the chosen method:
- Fishless Cycling: Generally, water should not be changed, as ammonia is the indispensable energy substrate for bacteria. However, “Substrate Inhibition” exists: if ammonia exceeds 5–8 ppm, the metabolism of Nitrosomonas is blocked by saturation. In this technical scenario, a water change is mandatory to “unblock” the cycle.
- Fish-in Cycling: In this case, water changes are imperative. The objective is to maintain toxicity levels below 0.5 ppm to avoid irreversible damage to the animals’ branchial epithelium.
The Fallacy of Crystal-Clear Water
It is vital to debunk the idea that water clarity is an indicator of biological health. The “Bacterial Bloom” phenomenon (cloudy or milky water), common in the second week, is a population explosion of heterotrophic, non-nitrifying bacteria.
Advice: Do not perform water changes for aesthetic reasons during a bacterial bloom. Intervening at this point disrupts the natural stabilization of the ecosystem. Allow the bloom to subside on its own as nutrients reach equilibrium.
Technological Trends in Biofiltration: The Future of Aquaria
The science of fishkeeping is not static; materials engineering is introducing solutions that complement and enhance the work of nitrifying bacteria. One of the most promising innovations is the integration of photocatalysis into biological support systems.
The Advancement of Cenospheres
Research by Snowdon et al. (2023) has marked a milestone by evaluating the efficacy of filters composed of Titanium Dioxide () cenospheres. This material proved to be an extremely effective oxidizing agent for mitigating the most critical nitrogenous contaminants:
- Nitrite () Reduction: A 55% decrease in nitrite concentrations was achieved compared to traditional control systems.
- Impact on Ammonia and Nitrates: The system reduced ammonia by 28% and nitrates by 17%, accelerating the chemical purification of the water.
Toward System Simplification
One of the most disruptive findings of this study is that a system composed exclusively of a biofilter and $TiO_2$ cenospheres exhibited performance equivalent to systems that include additional mechanical filtration. Both configurations showed identical results in critical water quality parameters, such as:
- Total Organic Carbon (TOC) and Chemical Oxygen Demand (COD).
- UV Absorbance (an indicator of organic purity).
- Fish physiological status, ensuring a safe and stable environment. This advancement suggests that, in the near future, reliance on sponges and frequent mechanical cleaning could be reduced through the use of intelligent, reactive filter media, allowing for low-maintenance aquaria with greater biological stability.
Conclusion: Cycling as Life Insurance
Aquarium cycling should not be perceived as a bureaucratic chore or a bothersome obstacle; it is, in essence, the life insurance for your fish. Whether you opt for a slow, natural biological protocol or implement technical acceleration strategies, the key to success will always lie in the triad: patience, measurement, and scientific rigor. It is imperative to distrust “24-hour magic solutions.” In professional fishkeeping, there is an inescapable maxim: only negative things happen quickly. By following this step-by-step guide, you are not just setting up a tank, but building a robust ecosystem where fauna will not only survive but thrive for years.
Toward a New Generation of Fishkeeping
As McKnight and Neufeld (2024) aptly conclude, a deep understanding of microbial ecology is fundamental to optimizing water treatment, both in the domestic sphere and the global aquaculture industry. These findings are charting the roadmap for the development of a new generation of biological supplements that incorporate comammox Nitrospira and AOA archaea, overcoming the limitations of current products based exclusively on AOB and NOB strains. Mastering the chemistry behind the glass is the first step toward becoming an elite aquarist. The future of biofiltration is already here, and it begins in your filter.
Aquarium Cycling Frequently Asked Questions (FAQ)
How long does it actually take to cycle an aquarium?
The standard natural process typically ranges from 4 to 8 weeks. However, this period can be reduced to 1 or 2 weeks through biological acceleration techniques such as “seeding” (using mature filter media) or inoculation with high-end bacteria that include Nitrospira strains.
Can I cycle my aquarium in just 24 hours with commercial products?
It is not recommended. Although “quick-start” products exist, studies such as Scagnelli et al. (2023) demonstrate that the majority fail to reduce ammonia within the first 14 days. The cycle is only considered complete when the system processes 2 ppm of ammonia to 0 ppm of nitrites within 24 hours, regardless of the product used.
Why did my aquarium water turn white or cloudy during the second week?
This phenomenon is known as a “Bacterial Bloom.” It is a population explosion of heterotrophic bacteria consuming nutrients within the water column. It does not indicate that the aquarium is cycled, nor is a water change necessary; the cloudiness will subside on its own once nutrients balance out and nitrifying bacteria establish themselves within the filter.
Is a water change necessary during cycling?
It depends on the method:
– Fishless Cycling: Water should not be changed unless ammonia exceeds 5–8 ppm (Substrate Inhibition).
– Fish-in Cycling: Daily water changes of 30–50% are mandatory to maintain ammonia levels below 0.25 ppm and prevent gill damage.
What is the difference between a “cycled” and a “mature” aquarium?
A cycled aquarium is chemically capable of processing nitrogen. A mature aquarium (after 3–6 months) has developed a complex biofilm and a stable microfauna. Biological maturity is what grants the ecosystem true resilience against diseases or imbalances.
Why do my plants stall the cycling process in the filter?
In a heavily planted aquarium (Silent Cycling), plants compete with bacteria for ammonia. If the plants absorb all the nitrogen, the filter’s bacterial colony will remain small. The risk is that if the plants die or are removed, the system may suffer a biological collapse due to a lack of backup bacterial infrastructure.
What is the best filter media for bacteria?
Current science, including studies by McKnight et al. (2025), suggests that materials with high Specific Surface Area (SSA), such as sintered glass or porous ceramic media, are ideal. Nitrospira bacteria and AOA archaea prefer these substrates to establish their biofilm colonies.
Why does the Walstad method discourage heavy aeration?
In the Walstad method, plants depend on the produced by the decomposition of organic matter within the substrate. Excessive aeration (via air stones) triggers a gaseous exchange that outgasses from the water, depriving plants of their primary carbon source and favoring the proliferation of algae.
References
Asadi, Sharif E. and Imanpure,Namin J. and Ramezanpoure,Z. The effect of media biofilter and aquatic plant (Ceratophyllum demersum) on water quality of recirculating aquaculture systems, growth and survival rates of ornamental fishes., (2022), English, Journal article, Iran, doi:10.22092/ijfs.2022.126393, 1562-2916, 21, (2), Tehran, Iranian Journal of Fisheries Sciences, (422–430), Iranian Fisheries Science Research Institute, )
Bagchi S, Vlaeminck SE, Sauder LA, Mosquera M, Neufeld JD, Boon N (2014) Temporal and Spatial Stability of Ammonia-Oxidizing Archaea and Bacteria in Aquarium Biofilters. PLoS ONE 9(12): e113515. https://doi.org/10.1371/journal.pone.0113515
Bik HM, Alexiev A, Aulakh SK, Bharadwaj L, Flanagan J, Haggerty JM, Hird SM, Jospin G, Lang JM, Sauder LA, Neufeld JD, Shaver A, Sethi A, Eisen JA, Coil DA. 2019. Microbial community succession and nutrient cycling responses following perturbations of experimental saltwater aquaria. mSphere 4: e00043-19. https://doi.org/10.1128/mSphere.00043-19.
Elmagzoub, W. A., Weidmann, M., Elnaiem, M. H. E., Dennig, A., Waller, U., Bernhard, A., Junhold, J., Abd El Wahed, A., Truyen, U., & Ceruti, A. (2026). Microbiome as a Tool to Monitor Aquarium Systems. Veterinary Sciences, 13(2), 125. https://doi.org/10.3390/vetsci13020125
Godzieba, M., Hliwa, P., & Ciesielski, S. (2025). Network of Nitrifying Bacteria in Aquarium Biofilters: An Unfaltering Cooperation Between Comammox Nitrospira and Ammonia-Oxidizing Archaea. Water, 17(1), 52. https://doi.org/10.3390/w17010052
McKnight MM, Neufeld JD.2024. Comammox Nitrospira among dominant ammonia oxidizers within aquarium biofilter microbial communities. Appl Environ Microbiol90:e00104-24.https://doi.org/10.1128/aem.00104-24
McKnight Michelle, Natasha Szabolcs, Alyssa Graham, Josh D Neufeld, Microbial community succession of home aquarium biofilters associated with early establishment of comammox Nitrospira, ISME Communications, Volume 5, Issue 1, January 2025, ycaf212, https://doi.org/10.1093/ismeco/ycaf212
Oliaro, F. J., Ajileye, O., George, I., Lamsal, S., Mosley, I. A., Ramirez, B., Sanders, T. L., Vanitshavit, V., Van Bonn, W., & Pinnell, L. J. (2025). The Role of Ammonia-Oxidizing Archaea During Cycling and Animal Introduction in a Newly Commissioned Saltwater Aquarium. Animals, 15(10), 1446. https://doi.org/10.3390/ani15101446
Ponpornpisit, Aranya; Jongjaroenjai, Malinee; Suthamnatpong, Naowarat; Piyapattanakorn, Sanit; and Burut-Archanai, Surachet (2024) “Single-tank sequencing batch bioreactor with a cellulose substrate for simultaneous nitrification and denitrification of an aquarium,” The Thai Journal of Veterinary Medicine: Vol. 53: Iss. 1, Article 8. DOI: https://doi.org/10.56808/2985-1130.3277
Sauder LA, Engel K, Stearns JC, Masella AP, Pawliszyn R, Neufeld JD (2011) Aquarium Nitrification Revisited: Thaumarchaeota Are the Dominant Ammonia Oxidizers in Freshwater Aquarium Biofilters. PLoS ONE 6(8): e23281. https://doi.org/10.1371/journal.pone.0023281
Scagnelli, A. M., Javier, S., Mitchell, M., & Acierno, M. (2023). Efficacy of quick-start nitrifying products in controlled fresh-water aquaria. Journal of Exotic Pet Medicine, 44, 22-26. https://doi.org/10.1053/j.jepm.2022.09.009
Snowdon, M. R., Liang, R. F. L., Kaur, A., Burton, E. A., Rathod, S., Fang, W., Dhiyebi, H. A., Bragg, L. M., Zhou, N. Y., Servos, M. R., & Freire-Gormaly, M. (2023). Effects of Substituting Activated Carbon with Titanium-Dioxide-Coated Cenospheres in Conventional Aquarium Filters. Environments, 10(11), 188. https://doi.org/10.3390/environments10110188
Walker, L., & Jiang, A. (2019). Dechlorination and nitrogen cycling at Georgia Aquarium for fresh and saltwater exhibits. Georgia Aquarium.
Zhou, J.S., Cheng, J.F., Li, X.D. et al. Unique bacterial communities associated with components of an artificial aquarium ecosystem and their possible contributions to nutrient cycling in this microecosystem. World J Microbiol Biotechnol 38, 72 (2022). https://doi.org/10.1007/s11274-022-03258-9
Editor at the digital magazine AquaHoy. He holds a degree in Aquaculture Biology from the National University of Santa (UNS) and a Master’s degree in Science and Innovation Management from the Polytechnic University of Valencia, with postgraduate diplomas in Business Innovation and Innovation Management. He possesses extensive experience in the aquaculture and fisheries sector, having led the Fisheries Innovation Unit of the National Program for Innovation in Fisheries and Aquaculture (PNIPA). He has served as a senior consultant in technology watch, an innovation project formulator and advisor, and a lecturer at UNS. He is a member of the Peruvian College of Biologists and was recognized by the World Aquaculture Society (WAS) in 2016 for his contribution to aquaculture.







