The clownfish (subfamily Amphiprioninae) is an iconic marine species, globally renowned for its symbiotic relationship with anemones. It is distinguished by its nature as a protandrous hermaphrodite—capable of undergoing a sex transition—its exceptional immunity to stinging toxins, and its high valuation within the ornamental species market. In response to the drastic decline in wild populations, aquaculture has emerged as a sustainable solution to meet global demand.
Whether you are a reef enthusiast seeking to recreate an oceanic ecosystem at home or a professional aquarist pursuing new challenges, this technical guide serves as the definitive resource for ensuring a thriving environment. Throughout this content, we will analyze everything from its evolutionary traits to the critical technical parameters for success in aquaculture and advanced fishkeeping.
From understanding their biological requirements to mastering technical maintenance, we will guide you through the complex care of these fascinating creatures. Discover the keys to configuring the ideal aquarium, providing balanced nutrition, and designing a harmonious habitat that precisely replicates their natural surroundings.
- 1 Key Takeaways: Essential Knowledge Regarding Clownfish
- 2 Species Classification and Diversity
- 3 Geographic Distribution and Natural Environment
- 4 Symbiosis: The Art of Coevolution
- 5 Nutrition and Feeding Strategies
- 6 Reproduction and Gender Dynamics
- 7 Maintenance and Water Quality Parameters
- 8 Compatibility and Coexistence Dynamics
- 9 Common Pathologies and Sanitary Management
- 10 Conclusion
-
11
Frequently Asked Questions (FAQ) Regarding Clownfish
- 11.1 Are all clownfish truly born male?
- 11.2 What is the ideal temperature to prevent malformations?
- 11.3 Is it mandatory to use Artemia to feed the larvae?
- 11.4 How can I intensify the color of my fish?
- 11.5 Why is my clownfish aggressive toward other fish?
- 11.6 What is the importance of darkness in egg hatching?
- 12 Bibliographic References
- 13 Entradas relacionadas:
Key Takeaways: Essential Knowledge Regarding Clownfish
- Transition Biology: They are protandrous hermaphrodites. The sex reversal from male to female is irreversible and governed by the ARCH model (Archetype, Rush, Context, and Phase)—a complex biological decision-making process rather than a merely linear progression.
- Symbiosis and Coevolution: The relationship with anemones is far from random. Fish select specific anemone lineages with which they have coevolved; for instance, the “bubble-tip” anemone (E. quadricolor) functions as a specialized species complex for various types of clownfish.
- Growth Optimization: Ideal cultivation temperatures range between 28°C and 30°C. Exceeding 31°C accelerates growth by 16%, yet reaching 34°C triggers the risk of skeletal malformations (such as lordosis) by up to 13.3%.
- Larviculture Revolution: Dependency on live Artemia can be eliminated. Implementing early weaning toward inert microdiets five days post-hatching can increase net profit by approximately $1.60 USD per fish.
- Color Enhancement: Supplementation with astaxanthin (minimum 0.5%) or natural extracts from Gac fruit and bell pepper is essential for the ornamental industry, achieving red pigmentation increases exceeding 75%.
- Water and Economic Efficiency: Juveniles can develop with a 95% survival rate at low salinities of 15‰. This brackish water strategy allows for savings of up to 57% in commercial salt costs.
- Welfare and Environment: Tank color significantly influences health. Utilizing dark-colored tanks instead of white ones drastically reduces cortisol (stress) levels in larvae, thereby improving their immune response to pathogens.
- Hatch Management: To ensure a successful and synchronized hatch, it is critical to guarantee absolute darkness and mechanical water agitation during the night of the eighth day post-spawning.
Species Classification and Diversity
Currently, 30 species of clownfish (also referred to as anemonefish) are recognized, distributed primarily within the genera Amphiprion and Premnas. Among the most iconic varieties are Amphiprion ocellaris (false percula), Amphiprion percula (orange clownfish), and Premnas biaculeatus (maroon clownfish), which are morphologically distinguished by their coloration patterns and dorsal spine counts.
While Amphiprion ocellaris is frequently confused with A. percula, recent studies have redefined our understanding of their lineage. According to Gaboriau et al. (2025), following the whole-genome sequencing of the 28 primary species, the most robust phylogenetic tree to date was established. This study reveals that Amphiprion biaculeatus represents a basal lineage for the rest of the group. Furthermore, the findings suggest that the common ancestor of these fish was an ecological generalist, and that specialization toward specific hosts (anemones) was an evolutionary process that occurred recurrently throughout their history.
In the cultural sphere, Amphiprion percula achieved worldwide fame through cinema; however, within the ornamental industry, Amphiprion ocellaris is the preferred species due to its remarkable resilience and ease of captive breeding.
Taxonomic Profile
The scientific classification of the subfamily Amphiprioninae is detailed below:
- Phylum: Chordata
- Class: Actinopterygii
- Clade: Percomorpha
- Family: Pomacentridae
- Genera: Amphiprion and Premnas
- Representative Species: A. percula, A. ocellaris, A. clarkii, P. biaculeatus.
- Common Nomenclature: Clownfish, Anemonefish.
Anatomy and Evolutionary Adaptations
The evolutionary lineage of the clownfish diverged approximately 35 million years ago, developing extraordinary biological adaptations that ensure their survival in reef environments:
- Ultraviolet (UV) Vision: They possess the ability to perceive the ultraviolet spectrum—a key evolutionary tool for identifying host anemones and recognizing other members of their social group.
- Acoustic Communication: They are capable of producing sounds by clashing their jaws. These vibrations are amplified by the swim bladder and are primarily used as warnings for territorial defense.
- Sexual Dimorphism and Size: A marked sexual dimorphism exists, wherein females exhibit a larger size than males. Depending on the species, their length can range between 7 cm and 17 cm.
Visually, these species are captivating. They present colorations ranging from vibrant yellow and orange to reddish and blackish hues, usually accented by white patches or bars. Understanding these morphological traits and the specific behaviors of each species will enable you to make an informed decision to guarantee a balanced ecosystem in your aquarium.
Stay Always Informed
Join our communities to instantly receive the most important news, reports, and analysis from the aquaculture industry.
Primary Species and Designer Varieties
Within the aquarium market, selecting the appropriate species is fundamental to the success of your tank:
Designer Clownfish Varieties
Due to advancements in selective aquaculture, unique morphological patterns are now available. Specimens such as the Orange Storm, Black Frostbite, and Snowflake are highly prized collector’s items in the contemporary ornamental industry.
“False Percula” Amphiprion ocellaris
This is the most popular specimen in aquaculture. It is characterized by possessing 11 dorsal spines and a highly attractive, brilliant orange coloration.

Amphiprion percula
Frequently confused with the ocellaris, it is distinguished by having thicker black margins on its white bands and 10 dorsal spines.

Amphiprion clarkii
Commonly known as the yellowtail clownfish (A. clarkii), females of this species can reach a length of 15 cm, whereas males typically only grow to 10 cm.

Premnas biaculeatus (Maroon Clownfish)
It is distinguished by its imposing size—reaching up to 17 cm—and a notably more territorial and aggressive temperament.

Geographic Distribution and Natural Environment
Clownfish are native to the tropical waters of the Pacific and Indian Oceans. Their distribution encompasses critical ecosystems such as the Red Sea, Australia’s Great Barrier Reef, Southeast Asia, the coasts of Japan, and the biodiverse Indo-Malaysian region. In these environments, their presence is intrinsically linked to the health of coral reefs and the availability of host anemones.
In the wild, these species exhibit one of the most fascinating social structures in the marine world. Groups are organized through a strict hierarchy based on size and dominance:
- Dominant Matriarchy: Communities are led by a female, who is the largest and most authoritative individual.
- Pair Bonding: They form exclusive breeding pairs. Following the female, the largest male holds the second rank in the social scale.
- Survival Roles: The male assumes the critical responsibility of defending the territory against intruders and ensuring sustenance for the female and offspring, thereby guaranteeing the viability of the clutch.
Understanding these natural dynamics is an indispensable requirement for any aquarist, as it allows for the replication of a harmonious environment that respects the biological instincts of these captivating creatures within the home aquarium.
Symbiosis: The Art of Coevolution
Mutualism and Chemical Protection
The relationship between the clownfish and the anemone represents one of the most perfect examples of mutualism in the marine kingdom. While contact with an anemone’s nematocysts (stinging cells) would prove lethal to other species, the clownfish has developed a specialized protective mucus that inhibits the tentacles’ discharge response, rendering the fish virtually “invisible” to its host. This bond offers bidirectional benefits: the anemone provides an impregnable refuge against predators, while the fish actively defends its host from parasites and butterflyfish, in addition to providing vital nutrients through its nitrogenous waste.
Classification by Specialization and Phenotype
Scientific research has delved into how this association shapes the appearance of the fish. According to Maison and Graham (2016), species such as A. percula primarily associate with Heteractis crispa, H. magnifica, and Stichodactyla gigantea. Furthermore, research by Gaboriau et al. (2025) reveals that clownfish have evolved into four morphological categories based on their reproductive habitat and specific host anemone:
- Generalists: Featuring dark bodies with two or three white bands; their morphology is versatile and similar to Entacmaea specialists.
- Radianthus magnifica Specialists: Possessing more elongated bodies, robust caudal peduncles, and a light orange coloration with reduced stripes.
- Entacmaea quadricolor Specialists: Characterized by a reddish-orange hue, a short snout, a deeper body, and thinner white markings.
- Stichodactyla Specialists: A highly differentiated evolutionary group adapted exclusively to this genus of anemones.
Recruitment and Genetic Selection
New evidence provided by Chiodo et al. (2026) suggests that the “bubble-tip” anemone (Entacmaea quadricolor) is not a single species, but rather a complex of diverse lineages whose evolution has been shaped by their symbionts. This study is crucial, as it demonstrates that the settlement of clownfish on the reef is not random; the fish “recruit” or specifically select anemone lineages with which they have coevolved for millennia.

Nutrition and Feeding Strategies
Ensuring a balanced diet is the fundamental pillar for the vitality and optimal growth of the clownfish. In its natural habitat, this species maintains an omnivorous diet, based primarily on zooplankton, small crustaceans, and vegetable matter (algae). In captivity, success lies in replicating this nutritional diversity through a multifaceted approach.
Guidelines for the Home Aquarium
To maintain the health of your specimens, a varied diet combining the following is recommended:
- Dry Foods: High-quality nutritional pellets and flakes.
- Fresh/Frozen Foods: Artemia, mysis shrimp, and seafood mixes.
- Frequency: It is preferable to provide small rations several times a day. This practice mimics their natural foraging behavior and prevents the degradation of water quality caused by excess waste.
Advancements in Aquaculture and Specialized Diets
Recent research has introduced critical components for optimizing professional breeding:
- Alternative Protein Sources: According to Vargas et al. (2019), the inclusion of insect meal (Hermetia illucens) is a sustainable alternative that does not compromise the growth or stress response of juveniles.
- Broodstock Nutrition: For Amphiprion ocellaris, Roux et al. (2021) suggest diets rich in polyunsaturated fatty acids, consisting of a blend of squid, mussels, shrimp, and nori seaweed.
- Color Optimization: Pigmentation is key in the ornamental market. While Díaz-Jiménez et al. (2021) recommend a minimum of 0.5% astaxanthin, a study by Tran et al. (2024) proposes the use of natural carotenoids (Gac fruit and bell pepper), achieving increases of up to 89% in red color intensity.
- Probiotic Health: Supplementation with Lactobacillus plantarum (10⁶ to 10⁸ CFU/g) significantly improves zootechnical performance and strengthens the immune system against pathogens such as Vibrio fluvialis (Paixão et al., 2020).
Larviculture: The Development Challenge
Historically, success in larval rearing has depended on the use of specialized live feed, such as rotifers (Brachionus plicatilis) and microalgae like Nannochloropsis oculata. However, recent research is transforming traditional protocols to maximize survival rates and reduce operational costs.
Advancements in Feeding and Weaning Protocols
Traditionally, authors such as Nass et al. (2016) suggested introducing Artemia nauplii as early as two days post-hatching for species like Amphiprion clarkii. Nevertheless, the study by Murray et al. (2025) on Amphiprion ocellaris represents a milestone by demonstrating that it is possible to drastically reduce dependence on costly live organisms.
The following details the most disruptive findings for the industry:
- Early Transition and Resilience: Larvae can transition from rotifers to Artemia at 5 days post-hatching (DPH) without compromising their total length or elevating whole-body cortisol levels (a key stress indicator).
- Elimination of the Artemia Phase: The most significant finding reveals that larvae can entirely bypass the Artemia stage if an inert microdiet is introduced promptly. Initiating the supply of commercial microdiets at 5 DPH ensures a superior survival rate (64%), outperforming the results of late weaning (8 or 11 DPH). It is important to note that inert feed should not be provided from day one, as initial survival strictly depends on the stimulus provided by live prey.
- Aesthetic Quality and Commercial Value: The choice of microdiet directly influences profitability. Utilizing feeds supplemented with astaxanthin (such as TDO Chroma Boost™) produces specimens with superior color saturation and brightness—decisive factors in the market value of ornamental fish.
Economic Impact on Aquaculture
The implementation of these new protocols optimizes not only the biology of the fish but also the producer’s finances. By eliminating the use of Artemia and transitioning directly from rotifers to a microdiet at 5 DPH, operational savings are estimated to translate into a net profit increase of approximately $1.60 USD per specimen produced.
Juvenile Nutrition and Pigmentation Optimization
Once the larval stage is surpassed, juvenile nutrition focuses on consolidating growth and, fundamentally, developing a competitive coloration for the ornamental market.
The Impact of Natural Astaxanthin
According to recent research by Hoffmann et al. (2025), strategic astaxanthin supplementation is the most effective method for enhancing the chromatic vitality of juvenile Amphiprion ocellaris. The key findings of this study include:
- Record-Breaking Results: Following a feeding period of just 60 days, a drastic improvement in red tone intensity was demonstrated. Scientists observed significantly more vibrant pigmentation in critical areas such as the ventral region and pectoral fins.
- Biological Efficiency: Utilizing natural sources from microalgae allows for superior aesthetic results within a shorter exposure time compared to standard diets, thereby optimizing captive production cycles.
- High-Availability Source: A dosage of 18 mg per kilogram of feed is recommended, specifically derived from the microalga Haematococcus pluvialis, which is renowned for its high concentration of natural pigments.
Reproduction and Gender Dynamics
The reproductive system of the clownfish is among the most unique mechanisms in the marine world. They are protandrous hermaphrodites: all individuals are born male and organize themselves into strict social hierarchies, where only the largest and most dominant specimen undergoes the biological transition to female.
The ARCH Model and Sex Reversal
Contrary to the popular belief that sex reversal is a simple, linear response to the absence of a female, a study by Rahman (2026) reveals a higher degree of complexity. This process is a “biological decision” governed by the ARCH model (Archetype, Rush, Context, and Phase). According to this multiplicative model, the transition is only triggered when specific hormonal and social thresholds are surpassed, ensuring that the reversal is successful and permanent.
In a typical anemone or aquarium, the structure is divided into:
- Alpha Female: The dominant and largest individual, responsible for egg-laying.
- Breeding Male: The second in rank and the only one charged with fertilization.
- Non-breeding Juveniles: Individuals whose development is stunted to avoid hierarchical conflict.
Breeding Protocols and Captive Management
Successful breeding in aquaria is possible if natural conditions are mimicked. Research by Lai et al. (2026) suggests precise parameters for the hybridization and rearing of Amphiprion ocellaris:
- Introduction Technique: To reduce aggression, the small male should be introduced first, followed by the female after 12 hours. This allows the male to establish his territory, facilitating the pair-bonding process.
- Optimal Dimensions: A size ratio of 2.5 ± 1 cm for males and 5 ± 1 cm for females is recommended.
The Life Cycle: From Egg to Hatching
The female clownfish, depending on the species, deposits between 600 and 1,500 eggs on a flat surface within the protection of the anemone. The male assumes the role of primary caregiver, taking responsibility for oxygenation and the removal of non-viable eggs.
A critical factor to consider is light pollution. According to Fobert et al. (2020), exposure to artificial light at night disrupts the reproductive lunar cycle, drastically reducing the hatching rate. Consequently, strict photoperiod control is essential for any aquaculture project.
Spawning Preferences and Environment
In the absence of natural anemones, the choice of substrate is a determining factor for success. According to Lai et al. (2026), species such as Amphiprion ocellaris exhibit a selective preference for rough-textured surfaces, such as ceramic tiles or hollow terracotta pots, while rejecting smooth or synthetic materials like PVC. This behavior suggests that broodstock prioritize environments that offer greater privacy and security for the clutch.
In terms of volume, studies on Amphiprion clarkii (Sarvi and Pourmozaffar, 2024) report batches of between 300 and 400 eggs, whereas the life cycle and larval rearing protocols for A. ocellaris have been extensively documented by Roux et al. (2019, 2021).

The Temperature Factor in Embryonic Development
Water temperature serves as the primary regulator of embryonic metabolism. Rodríguez-Ibarra et al. (2024) determined that while larval survival is optimal across a broad range, there are critical thresholds:
- Thermal Efficiency: The highest success rates are recorded at 30°C, achieving a 98.2% hatching rate and 96% survival.
- Developmental Velocity: Increased thermal levels accelerate the process; at 34°C, hatching occurs at 144 hours post-fertilization, compared to the 168 hours required at lower temperatures (26–30°C).
Synchronized Hatching Protocol
To maximize commercial outcomes, Yamanaka et al. (2025) recommend a specific protocol during the night of the eighth day following spawning:
- Mechanical Agitation: Water movement accelerates the digestion of the egg envelope (chorion), facilitating a rapid and, crucially, synchronized hatch.
- Absolute Darkness: This acts as the biological trigger for the secretion of hatching enzymes.
Clownfish Larviculture
Larval rearing represents the most demanding technical challenge in ornamental aquaculture. According to Tartila et al. (2023), this process requires rigorous management spanning from media preparation and egg stocking to the oversight of Recirculating Aquaculture Systems (RAS) and final harvesting.
Thermal Dynamics and Accelerated Growth
Temperature serves as the metabolic engine for larval development. Recent research presents key nuances for maximizing efficiency:
- Development and Morphology: Moore et al. (2023) demonstrated that A. ocellaris larvae reared at 31°C develop significantly faster, reaching critical stages in just 8 days and showing a 16% increase in length and ocular diameter compared to the standard.
- Biological Limits: Although Rodríguez-Ibarra et al. (2024) indicate that maximum growth occurs at 34°C, they warn that the ideal range to avoid malformations is situated between 28°C and 30°C.
- Species-Specific Optimization: For A. clarkii, Medeiros et al. (2026) recommend a combination of 30°C and a reduced salinity of 20‰. This finding is crucial for inland facilities, as it allows for a reduction in synthetic salt costs without compromising growth.
The Role of Parental Care and Digestive Physiology
Before hatching, male intervention is a determining factor. Arossa et al. (2025) report that parental care (fanning and rubbing) increases oxygen levels in the egg mass by 51%, protecting the embryos from local hypoxia.
Following hatching, the larvae exhibit surprising maturity. Sarvi and Pourmozaffar (2024) highlight that they possess a developed digestive system and a mouth gape of 250 µm, allowing them to consume standard rotifers immediately. The early formation of gastric glands (between days 11 and 15) facilitates an efficient transition toward artificial diets.
Ethology and Welfare: The Impact of Color
The physical environment directly influences hormonal health. A study by dos Santos et al. (2023) advises against using white tanks for rearing Amphiprion frenatus. It was discovered that larvae adapted to light backgrounds exhibit significantly higher cortisol (stress) levels in response to acute stimuli. The use of dark or neutral-colored tanks promotes a superior state of welfare and an improved immune response.
Once the larvae complete their metamorphosis, the management of juvenile specimens requires a profound understanding of their social instincts. Research by Wang et al. (2025) on Amphiprion ocellaris provides critical guidelines for the organization of captive populations.
Grouping Strategies and Survival
The study reveals that juveniles, regardless of their size (ranging from 2.0 cm to 4.0 cm), possess a high degree of sociality, showing a constant preference for remaining near their conspecifics. However, behavioral differences exist based on size:
- Small Juveniles (2.0–2.5 cm): They exhibit a much more pronounced tendency toward tight grouping, an evolutionary strategy linked to collective protection against potential predators.
- Large Juveniles (3.5–4.0 cm): Although social, they depend more on the stability of their environment and familiar groups to maintain low stress levels.
Recommendations for Grading
Based on these findings, it is suggested that the processes of grading (size-sorting) juvenile clownfish be performed early. Implementing these measures before the specimens reach larger sizes minimizes territorial conflicts and social stress. Performing the transition during the initial stages ensures a higher survival rate, as it avoids destabilizing the complex social bonds that larger fish establish with their environment.
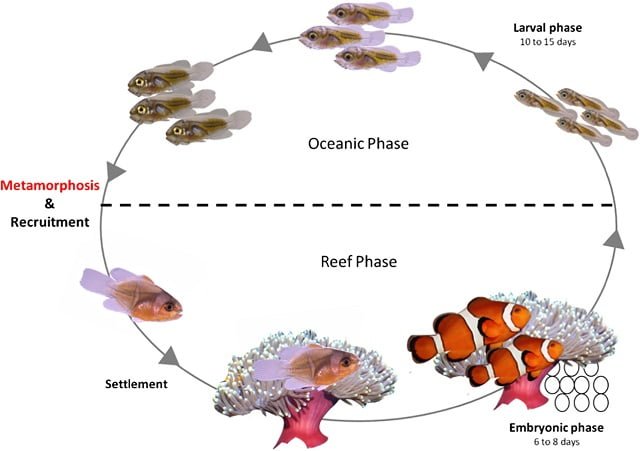
Maintenance and Water Quality Parameters
Long-term success in clownfish rearing depends on environmental stability. Maintaining optimal physicochemical parameters not only ensures survival but also maximizes growth and the overall profitability of the facility.
Stocking Density and Nutrition
For A. percula specimens, Chambel et al. (2015) established that the optimal density to achieve a superior condition factor is 2 fish per liter. Furthermore, it was determined that a diet containing 41% crude protein is the gold standard for promoting vigorous growth during the juvenile stage.
Temperature Management and Thermal Stress
While the tolerance range for A. ocellaris spans from 22°C to 30°C, the optimal temperature is situated at 26°C (Madeira et al., 2016). A critical finding by Versteeg et al. (2025) reveals that, when faced with marine heatwaves or extreme thermal stress, A. percula possesses the ability to “shrink” reversibly as a survival mechanism—a physiological response that underscores the importance of avoiding abrupt fluctuations.
Salinity Strategies and Operational Efficiency
The viability of brackish water rearing has opened new economic doors. Carneiro et al. (2024) confirmed that A. ocellaris juveniles can develop with a survival rate exceeding 95% at salinities as low as 15‰.
- Economic Benefit: This strategy allows for a reduction of up to 57% in commercial marine salt costs.
- Tolerance Limit: It is vital not to drop below 10 ppt, as according to Salleh et al. (2024), this is the species’ critical survival threshold.
Toxicity and Nitrogenous Compounds
Controlling ammonia and nitrites is the primary priority in Recirculating Aquaculture Systems (RAS). For the Maroon Clownfish (Premnas biaculeatus), unionized ammonia presents a lethality (LC50) of 0.89 mg/L at 96 hours (Rodrigues et al., 2014). Generally, it is recommended to maintain ammonia levels below 0.57 mg/L and nitrites below 25 mg/L to prevent tissue damage (Medeiros et al., 2019).
Technical Reference Sheet
The following table summarizes the essential values for managing a breeding system:
| Parameter | Recommended Range / Value |
| Aquarium Size (1♀ / 4♂) | Minimum 70 liters |
| Water Temperature | Optimal: 26°C (Range: 22 – 30°C) |
| Salinity (Specific Gravity) | 1.023 – 1.025 SG (or 15-35‰ depending on goal) |
| Water pH | 8.0 – 8.5 |
| Ammonia (NH₃) | 0 mg/L |
| Nitrites (NO₂) | < 0.1 mg/L |
| Photoperiod | 12h Light / 12h Darkness |
Compatibility and Coexistence Dynamics
The social behavior of clownfish is as fascinating as it is complex. Their communication system, based on acoustic stimuli (clicks), allows them to coordinate a highly organized internal hierarchy. Generally, these groups are composed of a dominant female, a breeding male, and a cohort of smaller juvenile individuals.
The Succession Mechanism
The stability of the group is guaranteed by its biological plasticity. According to the Great Barrier Reef Foundation, if the dominant female dies, the highest-ranking male initiates a permanent biological transition to become the new female. Simultaneously, the largest individual among the juveniles assumes the role of dominant male, ensuring the continuity of the reproductive cycle on the reef.
Territoriality and Aggression
In their natural habitat, anemonefish are fiercely territorial. They defend their host with surprising tenacity and rarely venture far from the protection of its tentacles. According to the Aquarium of the Pacific, aggression patterns—aimed at expelling intruders from their zone of influence—include:
- Tail-slapping.
- Constant chasing.
- Bites directed at the competitor’s head, abdomen, and fins.
Selecting Tank Mates
To ensure a peaceful environment in the home aquarium, the selection of companions must be based on temperament compatibility and similarity in environmental requirements. It is recommended to avoid excessively aggressive or large species that might compete directly for territory.
Recommended Tank Mates:
- Gobies and Blennies: Benthic species that typically occupy different zones of the aquarium.
- Small Wrasses: Active fish that share a compatible temperament.
Management Note: Regardless of the species, it is imperative to introduce new members gradually and monitor their interactions during the first few days to prevent acute territorial conflicts.
Common Pathologies and Sanitary Management
Prevention and early diagnosis are fundamental pillars for maintaining the health of captive clownfish populations. Below are the most prevalent viral and parasitic conditions reported in recent literature.
Amyloodiniosis (Velvet Disease)
This parasitic disease is one of the greatest threats in recirculating systems. Dhayanithi et al. (2022) have documented severe outbreaks of Amyloodiniosis specifically in the Orange Clownfish (A. percula).
- Treatment Protocol: Researchers recommend the therapeutic application of copper sulfate at a concentration of 10 ppm to effectively eradicate the parasite and control the spread of the outbreak.
Lymphocystis (LCDV)
Lymphocystis is a viral pathology affecting various species, with cases reported in A. percula (Lam et al., 2020), as well as in A. ocellaris and A. clarkii (Cheng et al., 2023).
- Clinical Signs: According to observations by Lam et al. (2020), affected specimens present characteristic white spots and wart-like growths on the dermis and fins.
- Diagnosis: Molecular investigations have confirmed the presence of the Lymphocystis Disease Virus (LCDV) in infected tissues, underscoring the importance of maintaining strict biosecurity protocols to prevent viral transmission between tanks.
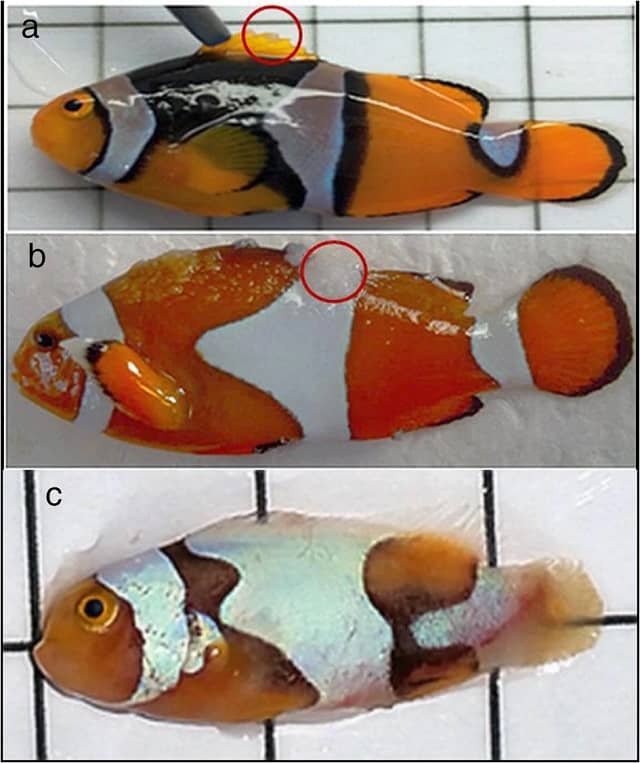
Malformations and Developmental Anomalies
Thermal control in aquaculture is a double-edged sword: while it accelerates metabolism, excessive temperatures can compromise the structural integrity of the specimens. Recent research has precisely quantified this risk.
The Impact of Temperature on Osteology
According to the findings of Rodríguez-Ibarra et al. (2024), there is a direct correlation between thermal increases and the incidence of skeletal deformities. The data reveal a critical safety threshold:
- Critical Incidence: The highest level of malformations (13.3%) is recorded in larvae cultured at 34°C.
- Safety Range: The percentage of anomalies drops drastically to 1.2% when the culture is maintained at a stable temperature of 28°C.
Typology of Deformities
The study details the most recurrent bone pathologies observed during larval development under thermal stress conditions:
- Lordosis: The most frequent deformity (11%), characterized by an abnormal inward curvature of the spine.
- Fin Anomalies: Malformations in the dorsal and anal structures reached 9.4%.
- Kyphosis: An outward curvature of the spine, present in 7.6% of cases.
- Scoliosis: Lateral deviations of the spinal column, recorded in 6.4% of the sample.
These results suggest that, to ensure high commercial and ethical production quality, breeders must prioritize thermal stability over growth speed.
Conclusion
Clownfish rearing is a profoundly rewarding endeavor that allows for the integration of ocean biodiversity into a controlled environment. By delving into their unique biological needs, ensuring a technically sound habitat, and maintaining impeccable water quality, you will be able to create an ecosystem where these iconic creatures do not merely survive but truly thrive and develop to their full potential.
From the strategic configuration of the aquarium and the design of a balanced diet to the precise selection of compatible tank mates, this technical guide has provided you with the scientific rigor and confidence necessary to successfully lead an aquaculture or home aquarium project. We invite you to immerse yourself in this fascinating challenge, applying the latest research advancements to create an underwater refuge that captivates the senses and guarantees the well-being of your specimens.
Frequently Asked Questions (FAQ) Regarding Clownfish
Are all clownfish truly born male?
Yes, clownfish are protandrous hermaphrodites. This means that all individuals are born with male reproductive organs. The transition to female only occurs due to a hierarchical necessity—such as when the dominant female dies—and is an irreversible biological process governed by the ARCH model.
What is the ideal temperature to prevent malformations?
Although they can tolerate broad ranges, 2024 studies indicate that the “sweet spot” for avoiding malformations is 28°C. While they grow faster at 34°C, the risk of skeletal deformities—such as lordosis—increases significantly, reaching up to 13.3%.
Is it mandatory to use Artemia to feed the larvae?
Not necessarily. According to research by Murray et al. (2025), it is possible to perform an early weaning to inert microdiets starting from 5 days post-hatching (DPH). This eliminates the dependency on Artemia, reducing operational costs without compromising survival rates.
How can I intensify the color of my fish?
The key lies in carotenoid supplementation. A minimum of 0.5% astaxanthin is recommended in the diet. Recent advancements suggest that utilizing natural extracts from Gac fruit or bell pepper can increase red color intensity by 75% to 89%.
Why is my clownfish aggressive toward other fish?
Aggression is a natural behavior linked to the defense of their territory and their host anemone. They utilize clicks to communicate and physical attacks—such as bites and constant chasing—to expel intruders. To minimize this, it is recommended to introduce the smaller male into the tank first.
What is the importance of darkness in egg hatching?
Absolute darkness acts as the biological trigger for embryos to secrete their hatching enzymes. Without this period of darkness, hatching may be delayed or become asynchronous, thereby compromising the overall viability of the clutch.
Bibliographic References
Aquarium of the Pacific. Clownfish: Amphiprion ocellaris
Arossa S, Steckbauer A, Klein SG, Parry AJ, Alva Garcia JV, Alamoudi T, Pluma N, Frappi S, Duarte CM. 2025 Oxygen availability modulates clownfish larvae responses to acute thermal stress in experimental settings. Proc. R. Soc. B 292: 20252201. https://doi.org/10.1098/rspb.2025.2201
Carneiro, M. D. D., Medeiros, R. S. d., Monserrat, J. M., Rodrigues, R. V., & Sampaio, L. A. (2024). Growth and Oxidative Stress of Clownfish Amphiprion ocellaris Reared at Different Salinities. Fishes, 9(1), 30. https://doi.org/10.3390/fishes9010030
Chambel, J., Severiano, V., Baptista, T. et al. Effect of stocking density and different diets on growth of Percula Clownfish, Amphiprion percula (Lacepede, 1802). SpringerPlus 4, 183 (2015). https://doi.org/10.1186/s40064-015-0967-x
Chiodo, T., De Jode, A., Quattrini, A. M., Gibson, M. K., Froehlich, C. Y., Huang, D., Fujii, T., Yanagi, K., Reimer, J. D., Scott, A., Rodríguez, E., & Titus, B. M. Nemo knows: Clownfishes differentiate cryptic host species across fine and broad geographic scales and reveal a diverse species complex in the clownfish-hosting sea anemones. Systematic Biology. https://doi.org/10.1093/sysbio/syag033
Cheng, M.-C.; See, M.S.; Wang, P.-C.; Kuo, Y.-T.; Ho, Y.-S.; Chen, S.-C.; Tsai, M.-A. Lymphocystis Disease Virus Infection in Clownfish Amphiprion ocellaris and Amphiprion clarkii in Taiwan. Animals 2023, 13, 153. https://doi.org/10.3390/ani13010153
Dhayanithi, N.B., Sudhagar, A., Kumar, T.T.A. et al. Study on amyloodiniosis outbreak in captive-bred percula clownfish (Amphiprion percula) and improved control regimens. J Parasit Dis 46, 1103–1109 (2022). https://doi.org/10.1007/s12639-022-01530-1
Díaz-Jiménez, L., Hernández-Vergara, M.P., Pérez-Rostro, C.I. and Olvera-Novoa, M.Á. (2021), The effect of two carotenoid sources, background colour and light spectrum on the body pigmentation of the clownfish Amphiprion ocellaris. Aquaculture Research, 52: 3052-3061. https://doi.org/10.1111/are.15149
dos Santos, T.M.L., de Oliveira Sousa, E.M., Tsuzuki, M.Y. et al. Tank color influences the response of tomato clownfish (Amphiprion frenatus) to an acute stress challenge. Fish Physiol Biochem 49, 577–584 (2023). https://doi.org/10.1007/s10695-023-01203-7
Fobert Emily K., Burke da Silva Karen and Swearer Stephen E. 2019. Artificial light at night causes reproductive failure in clownfish. Biol. Lett.152019027220190272
Gaboriau, T., Marcionetti, A., Garcia-Jimenez, A., Schmid, S., Fitzgerald, L. M., Micheli, B., Titus, B., & Salamin, N. (2025). Host use drives convergent evolution in clownfish. Proceedings of the National Academy of Sciences, 122(17), e2419716122. https://doi.org/10.1073/pnas.2419716122
Great Barrier Reef Foundation. Clownfish.
Hoffmann, H., Ozório, R.Á., Lopes, R.G. et al. Natural or synthetic carotenoid: impacts of Haematococcus pluvialis-derived astaxanthin and synthetic astaxanthin on the diet of clownfish Amphiprion ocellaris. Blue Biotechnol. 2, 17 (2025). https://doi.org/10.1186/s44315-025-00041-x
Lai, Y.-R., Sun, W.-T., Pan, C.-Y., Pan, C.-H., & Ding, D.-S. (2026). Evaluation of hybridization and spawning ground between Amphiprion ocellaris and Amphiprion ocellaris var. J. Fish. Soc. Taiwan, 53(3), 34-43. https://doi.org/10.29822/JFST.202609_53(3).0003
Lam, C., Khairunissa, I., Damayanti, L. et al. Detection of a new strain of lymphocystis disease virus (LCDV) in captive-bred clownfish Amphiprion percula in South Sulawesi, Indonesia. Aquacult Int 28, 2121–2137 (2020). https://doi.org/10.1007/s10499-020-00581-6
Madeira, C., Madeira, D., Diniz, M. S., Cabral, H. N., & Vinagre, C. (2016). Thermal acclimation in clownfish: an integrated biomarker response and multi-tissue experimental approach. Ecological Indicators, 71, 280-292.
Maison, K. A., & Graham, K. S. (2016). Status review report: orange clownfish (Amphiprion percula). NOAA.
Medeiros, R.S., Lopez, B.A., Sampaio, L.A. et al. Ammonia and nitrite toxicity to false clownfish Amphiprion ocellaris . Aquacult Int 24, 985–993 (2016). https://doi.org/10.1007/s10499-015-9965-9
Medeiros, A. F. F., Cruz, V. K., Carvalho, C. V. A., Owatari, M. S., & Tsuzuki, M. Y. (2026). Simultaneous impacts of salinity and temperature on the survival, growth and metamorphosis of yellowtail clownfish (Amphiprion clarkii) during larviculture. Int Aquat Res. https://doi.org/10.22034/iar.2026.2009738.1969
Moore, B., Jolly, J., Izumiyama, M., Kawai, E., Ryu, T., & Ravasi, T. (2023). Clownfish larvae exhibit faster growth, higher metabolic rates and altered gene expression under future ocean warming. Science of The Total Environment, 873, 162296. https://doi.org/10.1016/j.scitotenv.2023.162296
Murray, C. A., Lacy, B. D., Markham, O. I., & DiMaggio, M. A. (2025). Refining Larval Culture Protocols of Clownfish (Amphiprion ocellaris) to Reduce the Use of Live Feeds. Fishes, 10(9), 461. https://doi.org/10.3390/fishes10090461
Nass, D.H., Gonçalves, E.L.T. & Tsuzuki, M.Y. Effect of live food transition time on survival, growth and metamorphosis of yellowtail clownfish, Amphiprion clarkii, larvae. Aquacult Int 24, 1255–1261 (2016). https://doi.org/10.1007/s10499-016-9982-3
Paixão, P. E. G., do Couto, M. V. S., da Costa Sousa, N., Abe, H. A., Reis, R. G. A., Dias, J. A. R., … & Fujimoto, R. Y. (2020). Autochthonous bacterium Lactobacillus plantarum as probiotic supplementation for productive performance and sanitary improvements on clownfish Amphiprion ocellaris. Aquaculture, 526, 735395.
Rahman, T. (2026). Sex change in clownfish as an ARCH-governed biological decision. Hormones and Behavior, 180, 105907. https://doi.org/10.1016/j.yhbeh.2026.105907
Rodrigues, R.V., Romano, L.A., Schwarz, M.H., Delbos, B. and Sampaio, L.A. (2014), Acute tolerance and histopathological effects of ammonia on juvenile maroon clownfish Premnas biaculeatus (Block 1790). Aquac Res, 45: 1133-1139. https://doi.org/10.1111/are.12054
Rodríguez-Ibarra, Luz Estela, Abdo de la Parra, María Isabel, Velasco-Blanco, Gabriela, Aguilar-Zárate, Gabriela, & Martínez-Brown, Juan Manuel. (2024). Effect of temperature on the incubation, growth, survival, and presence of skeletal deformities in larvae of the clownfish Amphiprion ocellaris (Pomacentridae) under rearing conditions. Latin american journal of aquatic research, 52(3), 501-507. https://dx.doi.org/10.3856/vol52-issue3-fulltext-3151
Roux, N., Salis, P., Lambert, A., Logeux, V., Soulat, O., Romans, P., … & Laudet, V. (2019). Staging and normal table of postembryonic development of the clownfish (Amphiprion ocellaris). Developmental Dynamics, 248(7), 545-568.
Roux, N, Logeux, V, Trouillard, N, et al. A star is born again: Methods for larval rearing of an emerging model organism, the False clownfish Amphiprion ocellaris. J Exp Zool (Mol Dev Evol). 2021; 336: 376– 385. https://doi.org/10.1002/jezb.23028
Salleh, K. M., Lokman, E. D., Ma’mur, D. B., Ibrahim, N. S., & Yahaya, M. Z. (2024). Effect of salinity variation on the survival rate of Amphiprion ocellaris (Common Clownfish). INTERNATIONAL JOURNAL OF RESEARCH, 8, 455-466.
Sarvi, B., & Pourmozaffar, S. (2024). A review of the reproduction and rearing of clownfish, Amphiprion clarkii, in captivity. International Journal of Aquatic Research and Environmental Studies, 4(2), 145-158.
Tartila, S. S. Q., Abdillah, A. A., & Saramoutia, A. (2023). The Clownfish (Amphiprion spp.) Larviculture Technique with Recirculating Aquaculture System (RAS) in Buleleng, Bali. Journal of Aquaculture Development and Environment, 6(1), 363-369.
Tran DV, Luong HT, Pham KT, Dang TT, Hua NT, Pham HQ. Plant-based carotenoid supplementation: Growth, feed utilization efficiency, and coloration in false clownfish (Amphiprion ocellaris). Israeli Journal of Aquaculture – Bamidgeh. 2024;76(1). doi:10.46989/001c.94193
Vargas-Abúndez, A. J., Randazzo, B., Foddai, M., Sanchini, L., Truzzi, C., Giorgini, E., … & Olivotto, I. (2019). Insect meal based diets for clownfish: Biometric, histological, spectroscopic, biochemical and molecular implications. Aquaculture, 498, 1-11.
Versteeg, M. A., MacDonald, C., Bennett-Smith, M. F., Buston, P. M., & Rueger, T. (2025). Individual clown anemonefish shrink to survive heat stress and social conflict. Science Advances. https://doi.org/adt7079
Wang, G., Liu, J., Yang, J., Ma, S., Wang, Z., He, Y., Li, X., Yin, W., Li, X., Li, J., Xu, K., Wang, C., & Xu, W. (2025). Examination of Social Behavior and Cognition in Clownfish (Amphiprion ocellaris): Relationship to Artificial Rearing of Juveniles. Fishes, 10(11), 549. https://doi.org/10.3390/fishes10110549
Yamanaka, S., Okada, Y., Furuta, T., & Kinoshita, M. (2021). Establishment of culture and microinjection methods for false clownfish embryos without parental care. Development, Growth & Differentiation, 63, 459– 466. https://doi.org/10.1111/dgd.12759
Yamanaka, S., Kawaguchi, M., Yasumasu, S., Sato, K. and Kinoshita, M. (2025), Effects of Light and Water Agitation on Hatching Processes in False Clownfish Amphiprion ocellaris. Journal of Experimental Zoology Part B: Molecular and Developmental Evolution, 344: 29-40. https://doi.org/10.1002/jez.b.23276
Editor at the digital magazine AquaHoy. He holds a degree in Aquaculture Biology from the National University of Santa (UNS) and a Master’s degree in Science and Innovation Management from the Polytechnic University of Valencia, with postgraduate diplomas in Business Innovation and Innovation Management. He possesses extensive experience in the aquaculture and fisheries sector, having led the Fisheries Innovation Unit of the National Program for Innovation in Fisheries and Aquaculture (PNIPA). He has served as a senior consultant in technology watch, an innovation project formulator and advisor, and a lecturer at UNS. He is a member of the Peruvian College of Biologists and was recognized by the World Aquaculture Society (WAS) in 2016 for his contribution to aquaculture.






