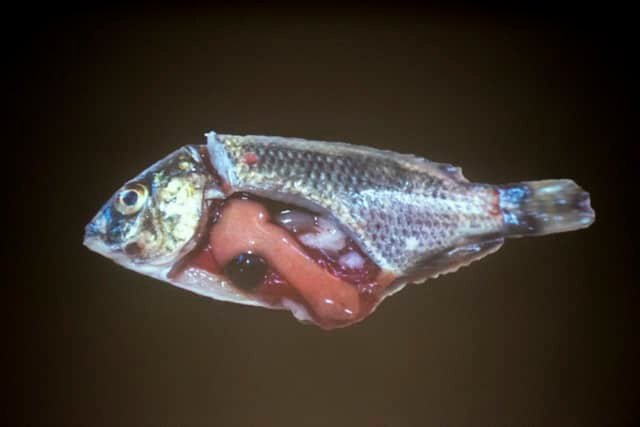
Tilapia Lake Virus (TiLV) is a deadly disease affecting tilapia or red tilapia aquaculture. TiLV causes mortality rates of up to 90% in affected fish populations.
TiLV affects tilapia at all stages of life, including fertilized eggs, larvae, fingerlings, juveniles, and adults. Vertical transmission from parents to offspring has been reported.
Clearing pathogenic viruses like TiLV during infection primarily depends on the presence of a functional immune system. In bony fish like tilapia, the innate immune system activates and triggers antiviral and pro-inflammatory responses during the early stages of infection.
Developing effective therapeutic and preventive strategies against viral diseases requires an understanding of the various processes and mechanisms of immunopathogenesis that occur during viral infections.
In this regard, scientists from the University College London, University of Veterinary Medicine Hannover, Moredun Research Institute, and the University of Nottingham published a scientific review on the mechanisms governing the antiviral response to TiLV infection, as it is crucial for the development of new drugs and antiviral treatment strategies.
Below is a brief summary of the study, which you can download at the end of the article.
TiLV Disease Pathology
The study highlights that the pathogenesis of TiLV is not yet fully defined and understood, partly because the cellular receptor for this virus has not been identified, and its mode of entry is still not completely resolved.
TiLV Tissue Tropism
TiLV appears to exhibit a very broad tissue tropism, as the virus can replicate in the brain, liver, kidneys, muscles, gills, fins, spleen, intestines, eyes, heart, ovaries, and testes of infected tilapia and zebrafish.
Infection Routes and Infection Models
TiLV seems to have a relatively narrow host range, with tilapia species being the canonical host of the virus. Therefore, immune responses to TiLV infection have been primarily measured in Nile tilapia and red hybrid tilapia.
Stay Always Informed
Join our communities to instantly receive the most important news, reports, and analysis from the aquaculture industry.
Innate Immune Response
TiLV appears to be well recognized by pattern recognition receptors (PRRs) that trigger various key host immune responses, such as increasing the release of antiviral factors important for restricting viral replication and spread.
In general, it is accepted that following viral infection, pathogen-associated molecular patterns (PAMPs) of the viruses, whether single-stranded or double-stranded unprotected RNA (dsRNA or ssRNA), are detected by cellular pattern recognition receptors (PRRs) located on the cell surface, in endosomes, or in the cytosol.
Adaptive Immunity to TiLV Infection
According to the study, the adaptive immune system recognizes foreign pathogens through two types of cellular receptors: the B cell receptor (BCR) and the T cell receptor (TCR). B and T cells are the main effector cells of the adaptive immune response.
The adaptive immune response is regulated by various mechanisms and increases with antigen exposure, producing immune memory, which forms the basis for vaccine development.
The adaptive response generally establishes itself days after infection and can recognize specific foreign antigens, leading to a response that increases in both speed and magnitude with subsequent exposures.
Knowledge Gaps
“It is clear that many questions remain to be addressed regarding the immune responses of tilapia to TiLV infection. The exact sites of virus entry remain to be determined, as well as the specific immune responses at those entry sites,” highlight the study’s authors.
They report that TiLV has been shown to persist in the brain for up to 90 days during infection. “Such prolonged viral persistence in the brain suggests that the brain is a privileged immunological site incapable of clearing the infection. Ideally, antibody development (especially neutralizing antibodies) during infection should provide sterile immunity against the pathogen,” they indicated.
The scientists point out that TiLV infection is lethal in most cases. However, there are reports of fish that survived the infection, suggesting the existence of specific immune mechanisms.
On the other hand, the researchers report that the specific mechanisms underlying the antiviral response to TiLV infection are still poorly studied and understood, which leaves:
The nature of the specific cells involved in innate immunity against TiLV infection, as well as the exact mechanisms by which TiLV suppresses and subverts the host immune response to establish infection, yet to be determined.
The complete spectrum of antibodies generated during TiLV infection is yet to be established.
The exact mechanisms of TiLV entry into the central nervous system (CNS), as well as the specific brain cells targeted by the virus, are yet to be discovered.
The study was funded by the Deutsche Forschungsgemeinschaft and the University of Veterinary Medicine Hannover Foundation.
Contacts
Japhette E. Kembou-Ringert
kjaphette@yahoo.fr
Mikolaj Adamek
mikolaj.adamek@tiho-hannover.de
Reference (open access)
Kembou-Ringert JE, Steinhagen D, Thompson KD, Daly JM and Adamek M (2023) Immune responses to Tilapia lake virus infection: what we know and what we don’t know. Front. Immunol. 14:1240094. doi: 10.3389/fimmu.2023.1240094
Editor at the digital magazine AquaHoy. He holds a degree in Aquaculture Biology from the National University of Santa (UNS) and a Master’s degree in Science and Innovation Management from the Polytechnic University of Valencia, with postgraduate diplomas in Business Innovation and Innovation Management. He possesses extensive experience in the aquaculture and fisheries sector, having led the Fisheries Innovation Unit of the National Program for Innovation in Fisheries and Aquaculture (PNIPA). He has served as a senior consultant in technology watch, an innovation project formulator and advisor, and a lecturer at UNS. He is a member of the Peruvian College of Biologists and was recognized by the World Aquaculture Society (WAS) in 2016 for his contribution to aquaculture.







