For millennia, agriculture has been the foundation of human civilization, nourishing bodies, fueling industries, and shaping landscapes. However, as our planet faces a growing population and the harsh realities of climate change, traditional agricultural practices are encountering unprecedented challenges.
Can we then envision a future where food security, resource sustainability, and environmental harmony coexist? The answer to this question lies in microalgae.
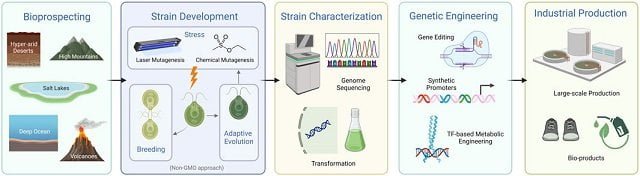
A team of scientists from the University of California San Diego published a scientific review highlighting innovative approaches aimed at overcoming existing barriers in microalgae cultivation. These approaches include a variety of techniques, from gene editing, synthetic promoters, and mutagenesis to selective breeding and metabolic engineering through transcription factors.
The Potential of Microalgae
Microalgae, found in oceans, lakes, and even hot springs, are not merely unicellular organisms; they are sustainability powerhouses. Forget vast expanses of fertile land and pristine water: microalgae thrive in uncultivable lands and brackish waters, challenging the limitations of traditional agriculture.
And their growth? Microalgae can multiply their biomass up to 20 times faster than terrestrial crops, a turning point for resource efficiency.
Nutritional Content
But the potential doesn’t end there. Microalgae are the “nutritional Swiss army knives” of nature. Packed with proteins, essential fatty acids like omega-3, and a rainbow of vitamins and minerals, they become not only an alternative food source but also a potent dietary supplement for both humans and animals alike. Imagine healthier fish in aquaculture, enhanced immunity in poultry, and improved nutrition in everyday food products, all thanks to these tiny titans.
Biofuel Production
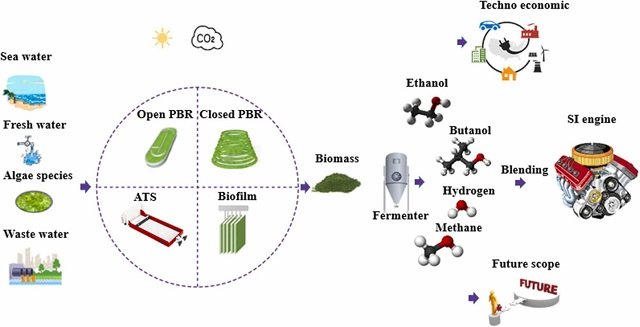
Beyond food, microalgae offer a green ray of hope for our energy woes. They can be efficiently converted into biofuels, a clean and renewable alternative to fossil fuels. And their diverse bioproducts don’t end there. From bioplastics and cosmetics to pharmaceuticals and nutraceuticals, microalgae are a true treasure trove of sustainable resources.
Carbon Capture
Microalgae are champions at carbon capture. They devour CO2, play a crucial role in mitigating climate change, and align perfectly with our environmental goals.
However, despite their immense potential, challenges persist. Large-scale and profitable biomass production remains an obstacle. But like true scientific adventurers, researchers are armed with ingenious tools to overcome this hurdle.
Stay Always Informed
Join our communities to instantly receive the most important news, reports, and analysis from the aquaculture industry.
Tools to Unlock the Potential of Microalgae
Scientists report that “various methods have been used to enhance biomass production, including better pond design, improved crop protection, better growth media, and water chemistry, enhancing photosynthetic efficiency, working with extremophile strains, and optimizing strain development through molecular engineering, breeding, selection, and in-vitro evolution.”
Gene editing, synthetic promoters, and mutagenesis are just some of the cutting-edge techniques employed to optimize microalgae growth and bioproduct performance. Imagine selectively cultivating microalgae for maximum efficiency or adjusting their metabolic pathways through transcription factors to produce specific bioproducts on demand.
Strategies for Designing Microalgae
According to the study, efforts to design microalgae primarily aim to strengthen their economic viability for bioproduct and biofuel production.
Additionally, scientists report the use of advanced genetic manipulation techniques to “fine-tune” the genome of many microalgae, making it easier to introduce specific traits that can significantly increase performance and reduce production costs.
The study provides a more detailed analysis of each of the strategies (bioprospecting, mutagenesis, genome sequencing, gene editing, and synthetic promoters), examining the comprehensive approaches used to optimize the productivity and resilience of microalgae for industrial applications.
Challenges in Microalgae Design
Various studies have highlighted that for microalgae cultivation, intended for bioproduct production, to achieve financial sustainability, it is crucial to innovate and implement cultivation strategies that can produce biomass at rates above 30 g/m2 per day.
To address these challenges, the study authors delve into the following topics:
- Working with extremophile microalgae and genetically designing them.
- Transcription factors and transcription factor binding sites in extremophile algae.
- Lipid and carbohydrate biosynthesis.
Conclusion
The scientific review has identified contemporary methods that enhance the quality of microalgae biomass, including gene editing and metabolic engineering, and acknowledges the challenges ahead, emphasizing the importance of focusing on commercially viable strains.
The future of food, feed, and fuels could lie in microalgae; however, challenges still need to be overcome. By harnessing the power of microalgae and continually innovating to overcome existing obstacles, we can unlock a sustainable future for generations to come.
The study has been funded by the United States Department of Energy.
Reference (open access)
Gupta, A., Kang, K., Pathania, R., Saxton, L., Saucedo, B., Malik, A., … & Mayfield, S. Harnessing Genetic Engineering to Drive Economic Bioproduct Production in Algae. Frontiers in Bioengineering and Biotechnology, 12, 1350722.
Editor at the digital magazine AquaHoy. He holds a degree in Aquaculture Biology from the National University of Santa (UNS) and a Master’s degree in Science and Innovation Management from the Polytechnic University of Valencia, with postgraduate diplomas in Business Innovation and Innovation Management. He possesses extensive experience in the aquaculture and fisheries sector, having led the Fisheries Innovation Unit of the National Program for Innovation in Fisheries and Aquaculture (PNIPA). He has served as a senior consultant in technology watch, an innovation project formulator and advisor, and a lecturer at UNS. He is a member of the Peruvian College of Biologists and was recognized by the World Aquaculture Society (WAS) in 2016 for his contribution to aquaculture.